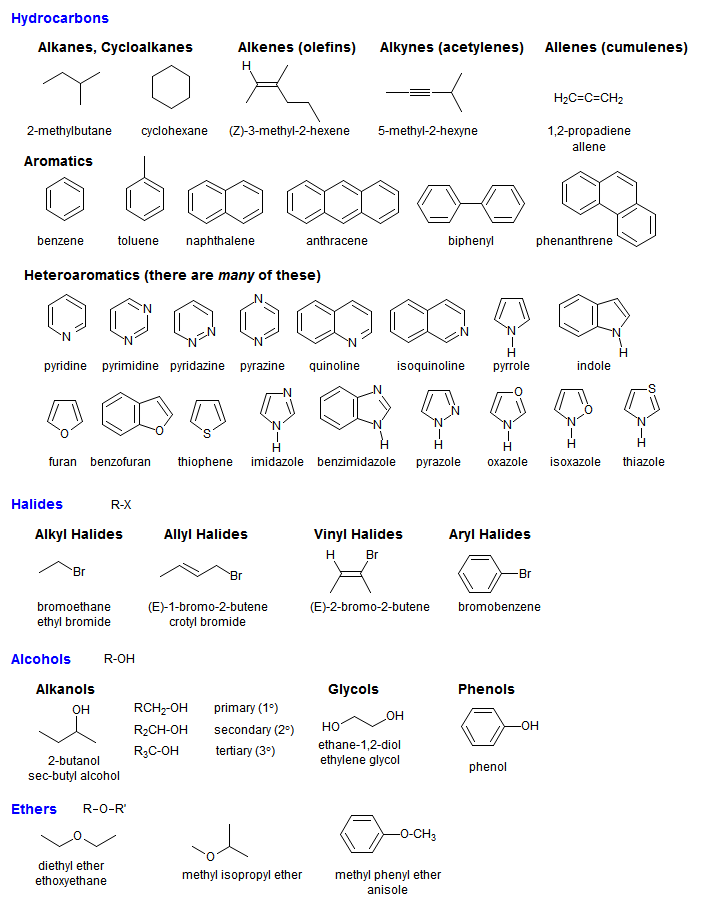
You should avoid using salt nomenclature for organic compounds that are not ethers, actual salts or derivatives of acids if convenient substituent names are available. Each of the words will be a radical name, not a compound name. If the last word ends in "ate" or "ide" you are using salt nomenclature, and your name will be at least two words (e.g.: butyl bromide, allyl iodide). In such names the "cation" and "anion" are always separate words, first the cation, then the anion (e.g.: sodium chloride, lithium hydride, cesium carbonate). Salt nomenclature was adopted from inorganic chemistry. Such names are one word, except for certain functional groups (notably acid derivatives like esters and acid chlorides) which retain their ancient two-word salt names. This system can be based on trivial names for the parent compound (chloroacetone) or on names based on systematic rules such as those developed by IUPAC or Chemical Abstracts. Substitution nomenclature takes a parent compound and identifies substituents which replace hydrogens on it with either prefixes (chloro-, phenyl-) or postfixes (-ol, -one). Here is a brief summary which should help keep things straight: The question of whether the name of an organic compound should be written in one word or two words is a perennial one.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
